The Origin of High Voltage Degradation in O3-Layered Oxide/Hard Carbon Sodium-Ion Pouch Cells
Z. Ye, L. Zhang, H. Hijazi, B. Tang, Y. Zhang, S. Yu, H. Ni, B. Rutherford, J. R. Dahn, and M. Metzger* “The Origin of High Voltage Degradation in O3-Layered Oxide/Hard Carbon Sodium-Ion Pouch Cells”, J. Electrochem. Soc. 173 010506 (2026). DOI: 10.1149/1945-7111/ae2f2e.
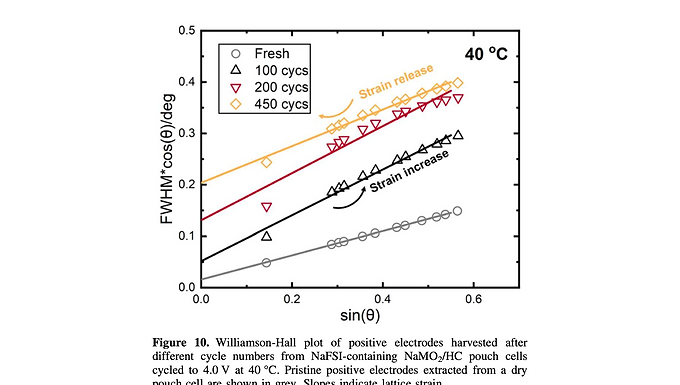
The O3-type sodium layered oxide NaMO2, where M is a mix of Mn, Fe, Ni and Zn, here NaMn0.39Fe0.31Ni0.22Zn0.08O2, has a specific capacity of ∼140 mAh g−1 when charged to 4.0 V, highlighting its potential as a positive electrode material for sodium-ion batteries. Electrochemical tests on machine-made pouch cells indicate that increasing the upper cut-off voltage from 3.8 to 3.9 V and 4.0 V leads to initially higher capacity which fades rapidly over cycling. Isothermal microcalorimetry, ultra-high precision coulometry and micro-X-ray fluorescence spectroscopy ruled out parasitic reactions between the electrolyte and the partially de-sodiated positive electrode surface as a major contributing factor to the inferior electrochemical performance at 4.0 V. Instead, it was found by ex situ and in situ X-ray diffraction that repeated cycling to 4.0 V induces the formation of an octahedral/prismatic hybrid phase, which leads to a collapse of the c lattice parameter in the crystal structure, resulting in poor long-term reversibility and ultimately poor cycle life. This irreversible structure transformation is the primary mechanism for poor cycling stability of the O3-type sodium layered oxide NaMO2 at high voltage.